
-
AB Members
Your journey to better hearing is always evolving. Learn how Advanced Bionics will be by your side for it all. Welcome to the AB family!
More
Your journey to better hearing is always evolving. Learn how Advanced Bionics will be by your side for it all. Welcome to the AB family!
MoreTesting has demonstrated that the Advanced Bionics HiRes implant family is MRI Conditional. Conditions vary by geography. A patient with the implant may be safely scanned with MRI only under very specific conditions. Scanning under different conditions may result in severe patient injury.
| Type of Implant | Model Number | MRI Field Strength (T) | Spatial Gradient Field (T/m) | Max Head SAR (W/kg) | Max Body SAR (W/kg) | |
|---|---|---|---|---|---|---|
| Clarion 1.0* | MMT-5100 L/R | MRI is contraindicated | None | None | None | |
| Clarion 1.2* | AB-5100 L/R | MRI is contraindicated | None | None | None | |
| Clarion CII Bionic Ear* | AB-5100H | MRI is contraindicated | None | None | None | |
| HiRes 90K* | CI-1400 | 1.5T | 2.5 T/m | ≤ 1 W/kg | ≤ 1.7 W/kg | |
| HiRes 90K Advantage | CI-1500 | 1.5T | 2.5 T/m | ≤ 1 W/kg | ≤ 1.7 W/kg | |
| HiRes Ultra | CI-1600 | 1.5T |
3.47 T/m | 13.90 T/m*** | ≤ 3.2 W/kg | ≤ 2 W/kg |
| 3.0T** | 6.9 T/m | ≤ 2.6 W/kg | ≤ 2 W/kg | |||
| HiRes Ultra 3D |
CI-1601 | 1.5T | 20 T/m | ≤ 3.2 W/kg | ≤ 2 W/kg | |
| 3.0T | 20 T/m | ≤ 2.6 W/kg | ≤ 2 W/kg | |||
* These devices are no longer sold in the EU or North America
** For MRI the magnet has to be removed surgically
*** With magnet removed

| Device | HiRes Ultra 3D |
|---|---|
| Instructions For Use |
HiResolution Bionic Ear System: HiRes Ultra 3D |
| 1.5T magnet in place |
Scanning ok under certain conditions |
| 3.0T magnet in place | Scanning ok under certain conditions |
The HiRes Ultra 3D cochlear implants have a specifically designed magnet that allows safe MRI scanning with the magnet in place, without angular restrictions of the head, surgical removal of the magnet or a bandaging protocol.
MRI Warnings
Do not allow patients with a HiRes cochlear implant to be in the area of an MRI
scanner unless the following conditions have been met:
NOTE: MRI procedures are contraindicated for CLARION (C1 and CII) cochlear implant recipients. For information regarding MRI use with HiRes 90K, HiRes 90K Advantage, and HiRes Ultra cochlear implants, please contact Advanced Bionics Technical Support.
NOTE: MRI safety was evaluated only for the HiRes Ultra 3D. Interactions between non-Advanced Bionics implants and the HiRes Ultra 3D during MRI are unknown.
Caution:
MRI Safety Information:
Testing has demonstrated that the HiRes Ultra 3D cochlear implant is MR Conditional. Unilateral and bilateral recipients with this device with magnet in place can be safely scanned in a horizontal closed bore quadrature coil MR system meeting the following conditions:
| MRI Field Strength | 1.5T | 3.0T |
|---|---|---|
| Maximum Spatial Field Gradient | 20 T/m | |
| RMS Gradient Field | 34.4 T/s | |
| Peak Slew Rate | 200 T/m/s | |
| Maximum whole body averaged SAR | 2.0 W/kg | 2.0 W/kg |
| Maximum head averaged SAR | 3.2 W/kg | 2.6 W/kg |
When tested under scan conditions defined above, the HiRes Ultra 3D cochlear implant produced a maximum temperature rise of <3˚C after 15 minutes of continuous scanning.
NOTE: During the scan, patients might perceive auditory sensations. Adequate counseling of the patient is advised prior to performing the MRI. The likelihood and intensity of the auditory sensations can be reduced by selecting sequences with a lower Specific Absorption Rate (SAR) and slower gradient slew rates.
The largest artifact at 3.0T is >15 cm when imaged under spin echo and gradient echo sequences. Smaller artifacts are possible using different scanning parameters only.
For additional information regarding the use of an MRI scanner with a HiRes Ultra 3D cochlear implant, please contact Advanced Bionics Technical Support at technicalservices@advancedbionics.com or visit www.advancedbionics.com/mri.

| Device | HiRes Ultra |
|---|---|
| Instructions For Use |
HiResolution Bionic Ear System: HiRes Ultra |
| 1.5T magnet removed |
Scanning ok under certain conditions |
| 1.5T magnet in place with MRI Antenna Coil Cover and bandaging protocol CI-7521 | Scanning ok under certain conditions |
| 3.0T magnet removed | Scanning ok under certain conditions |
For 1.5T MRI scans with the magnet in place
When tested under the scan conditions defined above the HiRes Ultra implant produced a maximum temperature of < 3° after 15 minutes of continuous 1.5T Scanning.
In MRI testing of unilateral recipient conditions, the image artifact caused by the device extends from the HiRes Ultra implant approximately 7.9 cm in a 1.5T MRI with the magnet in place and In MRI testing of bilateral recipient conditions >9.5 cm in a 1.5T MRI with the magnet in place using a spin echo or gradient echo pulse sequence.
These artifacts may result in a loss of diagnostic information in the implant vicinity.
Please note that the MRI Antenna Coil Cover CI-7521 and bandaging supplies must be on hand at the time of the MRI procedure.
The MRI Antenna Coil Cover is intended to be used along with a bandaging protocol to allow the patient to undergo an MRI procedure with the magnet left in place. Instructions for use must be followed, including the recommended bandaging protocol.
Compression Dressing prior to MRI Procedure with HiRes Ultra Users
This document will provide general guidance for applying appropriate compression dressing over the site of the implanted device (and magnet) in order to perform an MRI procedure
Hurt-Free Wrap – Hospital Grade

Flexible Tape

MRI Antenna Coil Cover CI-7521 - Advanced Bionics

Patient Headpiece

Marking Pen

Coach Self-Adhering Elastic Bandage 3 inch (8 cm) width

MRI Antenna Coil Cover CI-7521 - Advanced Bionics

Patient Headpiece

Marking Pen
1
Place patient in sitting position to allow access to the implant site.
NOTE: If the patient is bilaterally implanted, prepare to locate both implant devices using the following steps. Both implant locations must have compression dressing placed over their magnets.
2
Secure MRI Antenna Coil Cover over implant magnet site. Place the patient headpiece (with cable removed) over the implant site. The magnets will hold the headpiece in place.

Patient headpiece magnetically attached
3
Cut a piece of the Coach bandage that is long enough to wrap around the head once..
4
Wrap this piece around the head, so that the bandage covers the patient headpiece. Make this wrap tight.

Patient headpiece under bandage
5
Outline the position of the headpiece on the bandage using a marker or pen.

Marked bandage showing outline of headpiece
6
Slip out the patient headpiece, but keep the bandage in place.


Removing headpiece with bandage in place
7
Slide the MRI Antenna Coil Cover under the bandage, lining it up with the marked outline of the headpiece.

Inserting MRI Antenna Coil Cover

MRI Antenna Coin Cover under marked outline
8
Measure head size and bandage length needed for compression wrapping. Take the remaining Coach bandage roll, and wrap the bandage around the head once, without stretching.

Remaining Coach bandage roll wrapped once around head
9
Mark the location on the bandage that is one full wrap around the head. This is the head circumference.

Mark bandage one full wrap

Marked head circumference around head
10
Unwrap the piece of bandage with the marked head circumference, and place it on a flat surface.

Marked head circumference
11
Unroll the remaining bandage roll. Fold over the bandage start and crease at the marked head circumference.

12
Cut or tear the remaining bandage where it overlaps with the bandage start. The resulting bandage piece length is twice the head circumference.

13
Apply compression bandaging. Wrap this cut piece, this time very tightly, by stretching the marked line an additional half turn around the head.
NOTE: After 1.5 turns, the marked line should be on the opposite side of the head from where you started the wrap.
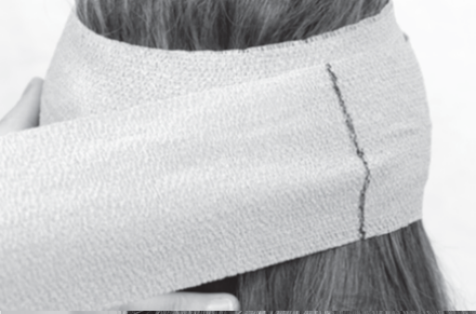
14
Continue wrapping the bandage at 150% extension for an additional 1.5 turns, resulting in 3 full turns total.
NOTE: After 3 turns, the bandage should end where you started the wrap (further is acceptable also).

Once the MRI procedure has been completed, gently remove the Coach Wrap, tape, and the MRI Antenna Coil Cover.
Discard all components including the MRI Antenna Coil Cover.
For 1.5T MRI scans with the magnet removed
When tested under the scan conditions defined above the HiRes Ultra implant produced a maximum temperature of < 3° after 15 minutes of continuous 1.5T Scanning.
In MRI testing of unilateral recipient conditions, the image artifact caused by the device extends from the HiRes Ultra implant approximately 3.1 cm in a 1.5T MRI with the temporary non-magnetic plug and In MRI testing of bilateral recipient conditions 4.2 cm in a 1.5T MRI with the temporary non-magnetic plug using a spin echo or gradient echo pulse sequence.
These artifacts may result in a loss of diagnostic information in the implant vicinity.
Please note that the MRI Antenna Coil Cover CI-7521 and bandaging supplies must be on hand at the time of the MRI procedure.
Magnet Removal/Replacement Using the HiRes Ultra Magnet Tool Kit, CI-1418
These tools must be sterilized prior to use. See the ”Guide for Reprocessing HiRes Ultra Reusable Tools” provided in the kit.
HiRes Ultra Magnet Took Kit, CI-1418

HiRes Ultra Magnet Tool Kit contains Two (2) Magnet Insertion Tools and Two (2) Magnet Pusher Tools
MRI Warning
3.0T MRI scans with the magnet removed
Magnet Removal/Replacement Using the HiRes Ultra Magnet Tool Kit, CI-1418
These tools must be sterilized prior to use. See the ”Guide for Reprocessing HiRes Ultra Reusable Tools” provided in the kit
HiRes Ultra Magnet Took Kit, CI-1418

HiRes Ultra Magnet Tool Kit contains Two (2) Magnet Insertion Tools and Two (2) Magnet Pusher Tools

| Device | HiRes 90k and HiRes 90K Advantage |
|---|---|
| Instructions For Use | HiResolution Bionic Ear System: HiRes 90K and 90K Advantage |
| 1.5T magnet removed | Scanning ok under certain conditions |
| 1.5T magnet in place with MRI Antenna Coil Cover and bandaging protocol CI-7521 | Scanning ok under certain conditions |
Magnetic Resonance Imaging Testing: MRI testing for the HiRes 90K Advantage cochlear implant, was performed according to two configurations:
with the internal magnet in place and with the internal magnet removed.
These tests were performed with MRI machines having a 1.5 Tesla static field, with a 64 MHz RF pulsed field, and a 0.3 Tesla static field, with a 12 MHz RF pulsed field, with the following results:
MRI is contraindicated except under the circumstances described below. Do not allow patients with a HiRes 90K or HiRes 90K Advantage cochlear implant to be in the area of an MRI scanner unless the following conditions have been met:
MRI testing performed for the HiRes 90K and HiRes 90K Advantage cochlear implant indicated that image shadowing may extend as far as 70 cm2 (with the magnet removed) and 210 cm2 (with the magnet in place) from the implant resulting in loss of diagnostic information in the implant vicinity. The extent of the shadowing may be minimized by adjusting the signal parameters.
MRI testing of the HiRes 90K and HiRes 90K Advantage cochlear implant with the internal magnet in place is only available in markets where regulatory approval has been received. Contact your Advanced Bionics representative for more information.
For additional information regarding the use of an MRI scanner with a HiRes 90K or HiRes 90K Advantage device, please contact Advanced Bionics Technical Support and please read the Magnetic Resonance Imaging (MRI) for the HiRes 90K and HiRes 90K Advantage CI (9050050269) or contact Advanced Bionics Technical Support.