
MRI Safety Information
For Advanced Bionics Cochlear Implants, Canada
Testing has demonstrated that the Advanced Bionics HiRes implant family is MRI Conditional. Conditions vary by geography. A patient with the implant may be safely scanned with MRI only under very specific conditions. Scanning under different conditions may result in severe patient injury.
MRI Safety Information by Type of Implant
| Type of Implant | Model Number | MRI Field Strength (T) | Spatial Gradient Field (T/m) | Max Head SAR (W/kg) | Max Body SAR (W/kg) | |
|---|---|---|---|---|---|---|
| Clarion 1.0* | MMT-5100 L/R | MRI is contraindicated | None | None | None | |
| Clarion 1.2* | AB-5100 L/R | MRI is contraindicated | None | None | None | |
| Clarion CII Bionic Ear* | AB-5100H | MRI is contraindicated | None | None | None | |
| HiRes 90K* | CI-1400 | 1.5T | 2.5 T/m | ≤ 1 W/kg | ≤ 1.7 W/kg | |
| HiRes 90K Advantage | CI-1500 | 1.5T | 2.5 T/m | ≤ 1 W/kg | ≤ 1.7 W/kg | |
| HiRes Ultra | CI-1600 | 1.5T |
3.47 T/m | 13.90 T/m*** | ≤ 3.2 W/kg | ≤ 2 W/kg |
| 3.0T** | 6.9 T/m | ≤ 2.6 W/kg | ≤ 2 W/kg | |||
| HiRes Ultra 3D |
CI-1601 | 1.5T | 20 T/m | ≤ 3.2 W/kg | ≤ 2 W/kg | |
| 3.0T | 20 T/m | ≤ 2.6 W/kg | ≤ 2 W/kg | |||
* These devices are no longer sold in the EU or North America
** For MRI the magnet has to be removed surgically
*** With magnet removed
Select the Implant name below for further MRI information
HiRes™ Ultra 3D Cochlear Implant



The external components of the HiResolution™ Bionics Ear System are MR Unsafe and must be removed before entering a room containing an MRI scanner.
The implant components of the HiRes™ Ultra 3D cochlear implant are MR conditional.
The HiRes Ultra 3D cochlear implants have a specifically designed magnet that allows safe MRI scanning with the magnet in place, without angular restrictions of the head, and without surgical removal of the magnet or a bandaging protocol.
Do not allow patients with a HiRes cochlear implant to be in the area of an MRI scanner unless the following conditions have been met:
- The external sound processor and headpiece must be removed before entering a room containing an MRI scanner.
- Verify that the implant, or both implants if bilaterally implanted, are compatible for conducting an MRI before proceeding. Failure to do so can lead to device movement, device damage, magnet movement, patient discomfort, or trauma and pain to the patient.
- For other MRI situations, i.e. if the recipient has two different types of Advanced Bionics Cochlear Implants (e.g. HiRes Ultra 3D and HR90k Advantage), the recipient must inform the MRI professional of the type of implants. If the recipient is not sure of the implant type, please contact the recipient’s cochlear implant clinic, the local AB office, or contact Advanced Bionics Technical Support at technicalservices@advancedbionics.com.
NOTE: MRI procedures are contraindicated for CLARION (C1 and CII) cochlear implant recipients. For information regarding MRI use with HiRes 90K, HiRes 90K Advantage, and HiRes Ultra cochlear implants, please contact Advanced Bionics Technical Support.
NOTE: MRI safety was evaluated only for the HiRes Ultra 3D. Interactions between non-Advanced Bionics implants and the HiRes Ultra 3D during MRI are unknown. - The recommended minimum duration of time post implant surgery prior to undergoing an MRI scan is 2 to 4 weeks in order to allow any inflammation to subside.
An MRI scan is not recommended if the patient has a fever.
CAUTION
- During the MRI procedure, you may experience pain, pressure, or discomfort. If this occurs, please notify your physician.
- Please consult with your physician prior to an MRI to determine if the benefits of an MRI are worthwhile over other imaging techniques.
- In case of two different implant types or models, MRI safety criteria that is most restrictive of the two implant types must be applied at the discretion of a qualified MRI professional.
Testing has demonstrated that the HiRes Ultra 3D cochlear implant is MR Conditional. Unilateral and bilateral recipients with this device with magnet in place can be safely scanned in a horizontal closed bore quadrature coil MR system meeting the following conditions:
MRI Field Strength |
1.5T |
3.0T |
|---|---|---|
| Maximum Spatial Field Gradient | 20 T/m | 20 T/m |
| RMS Gradient Field | 34.4 T/s | 34.4 T/s |
| Peak Slew Rate | 200 T/m/s | 200 T/m/s |
| Maximum Whole Body Averaged SAR | 2.0 W/kg | 2.0 W/kg |
| Maximum Head Averaged SAR | 3.2 W/kg | 2.6 W/kg |
When tested under scan conditions defined above, the HiRes Ultra 3D cochlear implant produced a maximum temperature rise of <3˚C after 15 minutes of continuous scanning.
Note: During the scan, patients might perceive auditory sensations. Adequate counseling of the patient is advised prior to performing the MRI. The likelihood and intensity of the auditory sensations can be reduced by selecting sequences with a lower Specific Absorption Rate (SAR) and slower gradient slew rates.
The largest artifact at 3.0T is > 15 cm when imaged under spin echo and gradient echo sequences. Smaller artifacts are possible if using different scanning parameters or a non-magnetic plug.
Note: For cases that would clinically benefit from reduced device artifact (for example, some head or neck scans), the internal magnet is surgically removed and possibly replaced with a Temporary Non-Magnetic Plug before the recipient undergoes an MRI procedure. The HiRes Ultra 3D cochlear implant can withstand 5 replacement cycles.
For additional information regarding the use of an MRI scanner with a HiRes Ultra 3D Cochlear Implant, please contact Advanced Bionics Technical Support at technicalservices@advancedbionics.com.
HiRes™ Ultra Cochlear Implant
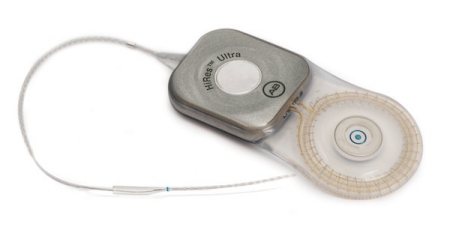
MRI Information
| Device | HiRes Ultra |
|---|---|
| Instructions For Use | HiResolution Bionic Ear System: HiRes Ultra |
| 1.5T magnet removed | Scanning ok under certain conditions |
| 1.5T magnet in place with MRI Antenna Coil Cover and bandaging protocol CI-7521 | Scanning ok under certain conditions |
| 3.0T magnet removed | Scanning ok under certain conditions |
MRI Warning
Do not allow patients with a HiRes Ultra cochlear implant to be in the area of an MRI scanner unless the following conditions have been met:
The bandaging protocol [Refer to the MRI Antenna Coil Cover (CI-7521) IFU for details] recommended by Advanced Bionics is followed when the patient undergoes an MRI procedure with the magnet left in place, or
For cases that would clinically benefit from reduced device artifact (for example some head or neck scans), the internal magnet is surgically removed and possibly replaced with the temporary nonmagnetic plug before the patient undergoes an MRI procedure.
The external sound processor and headpiece are MR Unsafe and must be removed before entering a room containing an MR scanner.
The recommended minimum duration of time post implant surgery prior to undergoing an MRI scan is 2 to 4 weeks in order to allow any inflammation to subside.
An MRI scan is not recommended if the patient has a fever.
For additional information regarding the use of an MRI scanner with a HiRes Ultra device, please contact Advanced Bionics Technical Support.
For 1.5T MRI Scans with the Magnet in Place
- Horizontal closed bore scanners with a static magnetic field of 1.5T (magnet in place).
- Maximum MR system reported, whole body averaged specific absorption rate (SAR) of ≤ 2 W/kg at 1.5T for quadrature transmit RF body coils.
- Maximum MR system reported, head averaged SAR of ≤ 3.2 W/kg at 1.5T for quadrature transmit RF head coils.
- RMS gradient field of 30 T/s and peak gradient field of 150 T/s.
- Maximum spatial field gradient of 3.47T/m at 1.5T (magnet in place)
When tested under the scan conditions defined above the HiRes Ultra implant produced a maximum temperature of < 3° after 15 minutes of continuous 1.5T Scanning.
In MRI testing of unilateral recipient conditions, the image artifact caused by the device extends from the HiRes Ultra implant approximately 7.9 cm in a 1.5T MRI with the magnet in place.
In MRI testing of bilateral recipient conditions >9.5 cm in a 1.5T MRI with the magnet in place using a spin echo or gradient echo pulse sequence.
These artifacts may result in a loss of diagnostic information in the implant vicinity.
Please note that the MRI Antenna Coil Cover CI-7521 and bandaging supplies must be on hand at the time of the MRI procedure.
MRI Warning
Do not allow patients with a HiRes Ultra cochlear implant to be in the area of an MRI scanner unless the following conditions have been met:
The bandaging protocol [Refer to the MRI Antenna Coil Cover (CI-7521) IFU for details] recommended by Advanced Bionics is followed when the patient undergoes an MRI procedure with the magnet left in place, or
For cases that would clinically benefit from reduced device artifact (for example some head or neck scans), the internal magnet is surgically removed and possibly replaced with the temporary nonmagnetic plug before the patient undergoes an MRI procedure.
The external sound processor and headpiece are MR Unsafe and must be removed before entering a room containing an MR scanner.
The recommended minimum duration of time post implant surgery prior to undergoing an MRI scan is 2 to 4 weeks in order to allow any inflammation to subside.
An MRI scan is not recommended if the patient has a fever.
For additional information regarding the use of an MRI scanner with a HiRes Ultra device, please contact Advanced Bionics Technical Support.
For 1.5T MRI Scans with the Magnet Removed
- Horizontal closed bore scanners with a static magnetic field of 1.5T (magnet removed).
- Maximum MR system reported, whole body averaged specific absorption rate (SAR) of ≤ 2 W/kg at 1.5T for quadrature transmit RF body coils.
- Maximum MR system reported, head averaged SAR of ≤ 3.2 W/kg at 1.5T for quadrature transmit RF head coils.
- RMS gradient field of 30 T/s and peak gradient field of 150 T/s.
- Maximum spatial field gradient of 13.90 T/m at 1.5T with magnet removed
When tested under the scan conditions defined above the HiRes Ultra implant produced a maximum temperature of < 3° after 15 minutes of continuous 1.5T Scanning.
In MRI testing of unilateral recipient conditions, the image artifact caused by the device extends from the HiRes Ultra implant approximately 3.1 cm in a 1.5T MRI with the temporary non-magnetic plug and In MRI testing of bilateral recipient conditions 4.2 cm in a 1.5T MRI with the temporary non-magnetic plug using a spin echo or gradient echo pulse sequence.
These artifacts may result in a loss of diagnostic information in the implant vicinity.
Please note that the MRI Antenna Coil Cover CI-7521 and bandaging supplies must be on hand at the time of the MRI procedure.
Magnet Removal/Replacement Using the Hires Ultra Magnet Tool Kit, Ci-1418
These tools must be sterilized prior to use. See the ”Guide for Reprocessing HiRes Ultra Reusable Tools” provided in the kit.
MRI Warning
The internal magnet is surgically removed and possibly replaced with the temporary non-magnetic plug before the patient undergoes an MRI procedure.
Do not allow patients with a HiRes Ultra cochlear implant to be in the area of an MRI scanner unless the following conditions have been met:
The bandaging protocol [Refer to the MRI Antenna Coil Cover (CI-7521) IFU for details] recommended by Advanced Bionics is followed when the patient undergoes an MRI procedure with the magnet left in place, or
For cases that would clinically benefit from reduced device artifact (for example some head or neck scans), the internal magnet is surgically removed and possibly replaced with the temporary nonmagnetic plug before the patient undergoes an MRI procedure.
The external sound processor and headpiece are MR Unsafe and must be removed before entering a room containing an MR scanner.
The recommended minimum duration of time post implant surgery prior to undergoing an MRI scan is 2 to 4 weeks in order to allow any inflammation to subside.
An MRI scan is not recommended if the patient has a fever.
For additional information regarding the use of an MRI scanner with a HiRes Ultra device, please contact Advanced Bionics Technical Support.
3.0T MRI Scans with the Magnet Removed
- Horizontal closed bore scanners with a static magnetic field of 3.0T (magnet removed)
- Maximum MR system reported, whole body averaged specific absorption rate (SAR) of ≤ 2 W/kg at 3.0T for quadrature transmit RF body coils.
- Maximum MR system reported, head averaged SAR of ≤ 2.6 W/kg at 3.0T for quadrature transmit RF head coils.
- RMS gradient field of 30 T/s and peak gradient field of 150 T/s.
- Maximum spatial field gradient of 6.90T/m at 3.0T with magnet removed
When tested under the scan conditions defined above the HiRes Ultra implant produced a maximum temperature of < 3° after 15 minutes of continuous 3.0T Scanning.
In MRI testing of unilateral recipient conditions, the image artifact caused by the device extends from the HiRes Ultra implant approximately 4.7 cm in a 3.0T MRI with the temporary non-magnetic plug.
In MRI testing of bilateral recipient conditions 4.7 cm in a 3.0T MRI with the temporary non-magnetic plug using a spin echo or gradient echo pulse sequence.
These artifacts may result in a loss of diagnostic information in the implant vicinity.
Please note that the MRI Antenna Coil Cover CI-7521 and bandaging supplies must be on hand at the time of the MRI procedure.
Magnet Removal / Replacement Using the Hires Ultra Magnet Tool Kit, Ci-1418
These tools must be sterilized prior to use. See the ”Guide for Reprocessing HiRes Ultra Reusable Tools” provided in the kit.
HiRes™ 90K Cochlear Implant Family

MRI Information
| Device | HiRes 90k and HiRes 90K Advantage |
|---|---|
| Instructions For Use | HiResolution Bionic Ear System: HiRes 90K and 90K Advantage |
| 1.5T magnet removed | Scanning ok under certain conditions |
| 1.5T magnet in place with MRI Antenna Coil Cover and bandaging protocol CI-7521 | Scanning ok under certain conditions |
MRI Warning
The HiRes 90K implant family, with either the internal magnet removed or left in place, has been tested with 1.5 Tesla/64 MHz and 0.3 Tesla/12 MHz MRI systems.
MRI is contraindicated except under the circumstances described below.
Do not allow patients with an implant from the HiRes 90K family to be in the area of an MRI system unless:
- The bandaging protocol recommended by Advanced Bionics (029-M486) is followed when the patient undergoes an MRI procedure with the magnet left in place or,
- The internal magnet is surgically removed and possibly replaced with the Magnet Insert Dummy before the patient undergoes an MRI procedure.
- The external sound processor and headpiece are removed before entering a room where an MRI scanner is located.
MRI parameters should be selected to ensure a Specific Absorption Rate (SAR) of less than 1.0 W/kg in the head region at 1.5T.
Patient should be monitored verbally and visually throughout the MRI procedure.
Image shadowing may extend as far as 70 cm2 (without magnet) and 210 cm2 (with magnet) around the implant, resulting in loss of diagnostic information in the implant vicinity. The extent of the shadowing may be minimized by adjusting the signal parameters.
For additional information regarding the use of MRI, please read the Magnetic Resonance Imaging (MRI) for the HiRes 90K and HiRes 90K Advantage CI (9050050269) or contact Advanced Bionics Technical Support.
Note: In the EU, MRI Examination Request Form (029-M569) must be provided to Advanced Bionics for review and approval prior to performing the MRI.
